More than 70 countries, including Brunei and Malaysia, have included the Human Papilloma Virus (HPV) vaccine in their national immunisation programme. This effectively protects a good number of females against cervical cancer, which has a high mortality rate of almost half of those who get diagnosed. Cervical cancer vaccination, is hence a simple step, of of great importance as they have the potential to reduce the overall burden of cervical cancer.
What is Cervical Cancer?
It is a type of cancer that occurs in the lower part of the uterus known as the cervix that is connected to the vagina below. The main causative agent is a virus named Human papillomavirus. Human papillomavirus (HPV) is a sexually transmitted virus. Two strains – type 16 and type 18 are responsible for about 70% of all cervical cancer cases.
HPV causes anal, genital, and oropharyngeal disease in both males and females.
Risk Factors for Cervical Cancer
- A higher number of pregnancies
- Oral contraceptive use
- Low socioeconomic condition
- Multiple sexual partners
- Being sexually active at an earlier age
- Having other STIs — such as chlamydia, gonorrhea, syphilis, and HIV/AIDS
- Smoking
- Exposure to miscarriage prevention drugs such as diethylstilbestrol (DES)
- Compromised immunity
Signs & Symptoms of Cervical Cancer
Although symptoms are not pronounced in the early stage of the disease, the following clinical features may be seen in advanced stages of cervical cancer:
- Vaginal bleeding after intercourse, between periods, or after menopause
- Vaginal discharge that may be watery or bloody with a foul odour
- Lower abdominal pain
- Pain during sexual intercourse
The risk of developing cervical cancer can be prevented to a great extent by having screening tests and receiving a vaccine that protects against HPV infection.
Prevention with Cervical Cancer Vaccination
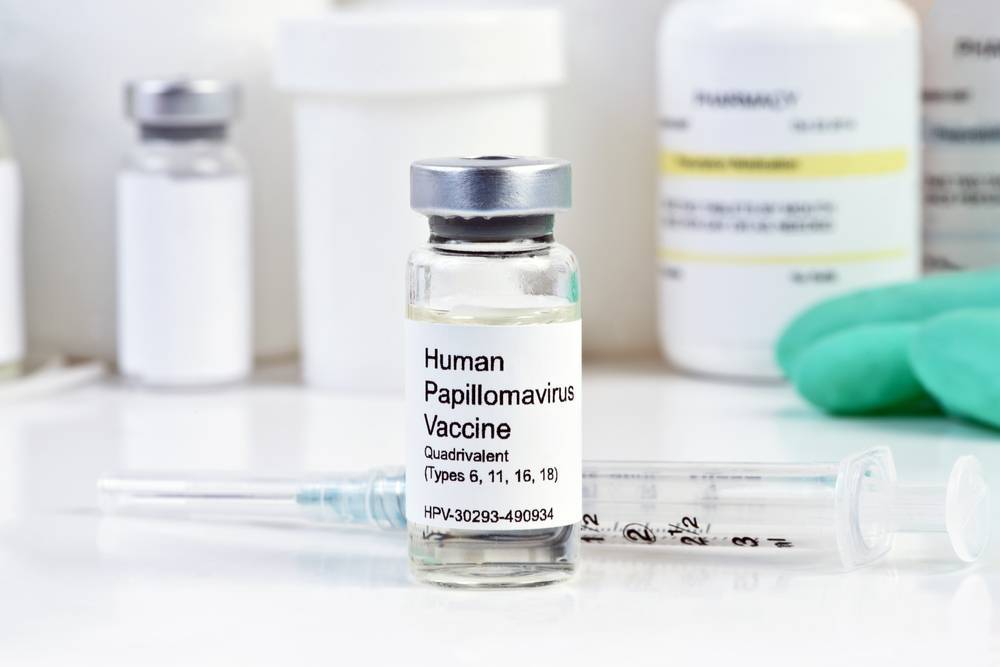
HPV vaccination can prevent more than 90% of cervical cancers and also protects against other infections and warts due to HPV. It contains proteins belonging to the virus, that stimulate the body’s immune system to produce antibodies against any subsequent attack of HPV. (Read more on how vaccinations work here)
There are currently 3 vaccines that have been approved for use. They are:
- 9-valent HPV vaccine (Gardasil 9)
- Quadrivalent HPV vaccine (Gardasil)
- Bivalent HPV vaccine (Cervarix)
9-valent HPV is the newest vaccine available in the USA and it protects against 9 types of viruses i.e. HPV 6, 11, 16, 18, 31, 33, 45, 52, 58.
Quadrivalent Gardasil contains a protein present in four types of HPV responsible for causing cancer in the cervix (types 6, 11, 16, and 18).
Cervarix is a bivalent vaccine i.e. it protects against only two types of HPV (16 and 18).
Cervical Cancer Vaccination Schedule
The World Health Organization (WHO) currently recommends 2 doses at an interval of 6 months for cervical cancer. The vaccination is mainly targeted at girls aged 11 to 14 years. But the vaccines are indicated for females as young as 9 years old.
The vaccine is most effective when given before the onset of sexual activity because the vaccines do not treat pre-existing HPV infections.
For individuals aged 15-26 years old, the HPV vaccine is administered in three doses at 0, at 1 to 2 months, and at 6 months. Similarly, an immunocompromised patient should also receive a three-dose schedule.
Vaccination in individuals aged 27 and older should be made on an individual basis. Because exposure to the HPV virus increases with age, the population benefit and cost-effectiveness of vaccination goes down. However, if the patient has no prior sexual experience or has had few sexual partners with low risk of HPV exposure, their choice to vaccinate may still be advantageous. It would still be best to check with your practitioner on the local guidelines and approved ages for vaccination in your country.
Side effects
Typical local reactions e.g. pain at injection site, redness and swelling, and fever may be experienced.
Who should not receive the Cervical Cancer Vaccination?
- Persons with a history of immediate hypersensitivity to yeast.
- A moderate or severe acute illness, with or without fever. The vaccine must not be given until symptoms of the acute illness improve.
- Pregnant women
- However, if found to be pregnant after completion of part of the vaccine, there’s no need to worry. The remainder of the vaccine schedule should be completed after completion of pregnancy.
Efficacy of the vaccine
Following mass vaccination programs in the Western countries, the Central disease control has stated that most HPV cancers and genital warts have dropped by 86 percent among teen girls. Similarly, the percentage of precancerous cervical lesions caused by the HPV types has also fallen by 40 percent. Cervical cancer vaccination can prevent more than 32,000 of these cancers from ever developing by preventing the infections that cause those cancers.
However, as with all vaccines, there is no 100% protection. Cervical cancer vaccination is also not a substitute for routine cervical cancer screening. Vaccinated women are still encouraged to go for their routine Pap tests:
- Once every 3 years for those between 25 – 29 years old
- Once every 5 years for those above 30 years old
References
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. [PubMed] [Google Scholar]
- Singh Y, Shah A, Singh M, Verma S, Shrestha BM, Vaidya P, Nakarmi RP, Shrestha SB. Human papillomavirus vaccination in Nepal: an initial experience in Nepal. Asian Pac J Cancer Prev. 2010;11(3):615-7. PMID: 21039025.
- World Health Organization. Global strategy to accelerate the elimination of cervical cancer as a public health problem
- Merck and Company. Prescribing Information for Gardasil. Accessed December 4, 2006.
- Virtual Mentor. 2007;9(1):30-33. doi: 10.1001/virtualmentor. 2007.9.1.cprl1-0701.
- Dangal G. Cervical Cancer and HPV Vaccine in Nepal: Issues and Challenges
- CDC. Human papillomavirus vaccination coverage among adolescent girls, 2007–2012, and postlicensure vaccine safety monitoring, 2006–2013—United States. MMWR. 2013;62:591–5. – PMC – PubMed
- HealthHub Singapore. HPV Prevention: HPV vaccination (Singapore). October 16,2020. [online] Accessed 27th April 2021.
- Bruni L, Barrionuevo-Rosas L, Albero G, et al. ICO Information Centre on HPV and Cancer (HPV Information Centre). Human papillomavirus and related diseases in the world: Summary report. December 15, 2016. http://www.hpvcentre.net/statistics/reports/XWX.pdf (Accessed on April 13, 2017).
Article written in conjunction with World Immunisation Week 2021.