
Study Finds Ozempic and Wegovy Safe: No Increased Risk of Suicidal Thoughts
Recent study reveals semaglutide, used in Ozempic & Wegovy, shows no increased suicidal ideation risk,
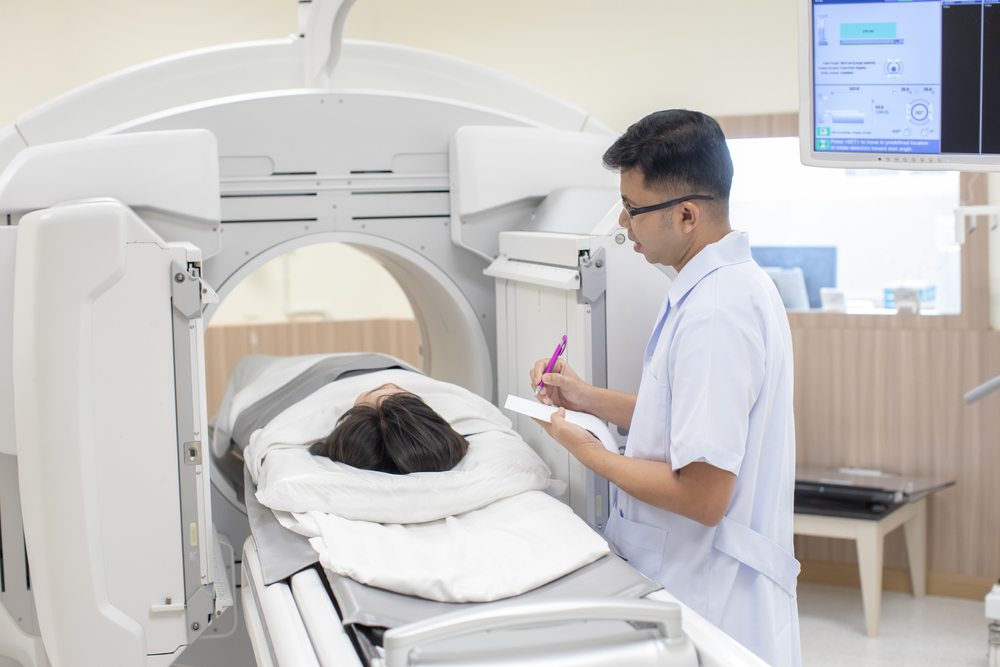

Recent study reveals semaglutide, used in Ozempic & Wegovy, shows no increased suicidal ideation risk,

Bad breath could be commonly experienced in the morning. However, it could also be a

Senator Christopher “Bong” Go, chairperson of the Senate Committee on Health and Demography, has expressed

Why do some people have cancer run in the family? It does not mean that it is directly passed down from parents to children.

As Palawan charts its course towards a malaria-free future, the unity and commitment displayed at the 14th Provincial Malaria Congress serve as a beacon of hope. With healthcare providers, officials, and the community working hand in hand, Palawan is poised to eliminate the disease, contributing significantly to the nationwide goal of a Philippines entirely free from this infectious disease. The efforts in Palawan, underscored by resilience and dedication, illuminate the path forward and exemplify the collective strength required to overcome the final challenges in this fight. The province’s journey serves as an inspiration for regions worldwide grappling with the persistent threat of mosquito-borne diseases.
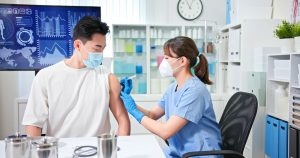
Find out who should get vaccinated for the flu and discover crucial information that can keep you and your loved ones safe.




